Valganciclovir
- Click on QUICK INQUIRY to receive a quote from our team of experts.
- With the quality product at a COMPETITIVE price, you can focus more on your research.
Overview
Description
Valganciclovir is an antiviral medication primarily used to treat cytomegalovirus infections, particularly in immunocompromised patients such as those with acquired immunodeficiency syndrome or following organ transplantation . It is the L-valyl ester of ganciclovir and acts as a prodrug, which means it is converted into its active form, ganciclovir, after administration . This compound is known for its effectiveness in preventing the spread of cytomegalovirus by inhibiting viral DNA synthesis .
Preparation Methods
Synthetic Routes and Reaction Conditions: Valganciclovir is synthesized through a multi-step process involving the esterification of ganciclovir with L-valine. The synthesis begins with the protection of ganciclovir, followed by esterification with L-valine using reagents such as dicyclohexylcarbodiimide and 4-dimethylaminopyridine . The final product is obtained through deprotection and purification steps .
Industrial Production Methods: Industrial production of this compound involves similar synthetic routes but on a larger scale. The process includes the use of high-performance liquid chromatography for purification and quality control . The production is optimized to ensure high yield and purity, adhering to stringent pharmaceutical standards .
Chemical Reactions Analysis
Types of Reactions: Valganciclovir undergoes hydrolysis to convert into its active form, ganciclovir . This hydrolysis is facilitated by intestinal and hepatic esterases . The compound does not undergo significant oxidation or reduction reactions under physiological conditions.
Common Reagents and Conditions: The hydrolysis of this compound to ganciclovir is catalyzed by esterases present in the liver and intestines . No additional reagents are required for this conversion.
Major Products Formed: The primary product formed from the hydrolysis of this compound is ganciclovir, which is the active antiviral agent .
Scientific Research Applications
Clinical Applications
1. Treatment of Cytomegalovirus Retinitis
Valganciclovir is effective in treating CMV retinitis, particularly in patients with HIV/AIDS. Studies have shown that it has comparable efficacy to intravenous ganciclovir while offering the advantage of oral administration, which reduces hospital visits and associated costs .
2. Prophylaxis in Solid Organ Transplant Recipients
In solid organ transplant recipients, this compound is utilized for both preemptive therapy and treatment of CMV disease. A study indicated that it can effectively prevent CMV infections post-transplant, reducing the incidence of CMV disease compared to no prophylaxis or other antiviral therapies .
3. Treatment in Pediatric Populations
Research has demonstrated that this compound is also effective in treating CMV infections in children, with pharmacokinetic studies indicating appropriate dosing adjustments based on age and renal function .
Pharmacokinetics and Pharmacodynamics
This compound's pharmacokinetics involve its conversion to ganciclovir, which is then metabolized and excreted by the kidneys. A recent population pharmacokinetic/pharmacodynamic model developed for kidney transplant patients showed that appropriate dosing can achieve significant viral load reductions within weeks of therapy initiation .
Case Studies
Case Study 1: Lung Transplant Recipient
A notable case involved a female lung transplant recipient who maintained this compound prophylaxis effectively through therapeutic drug monitoring. After experiencing complications post-surgery, she was able to achieve viral suppression with careful management of her this compound dosage .
Case Study 2: Solid Organ Transplant Efficacy
In a clinical trial involving solid organ transplant recipients, this compound was shown to be noninferior to intravenous ganciclovir for treating established CMV disease. The trial highlighted its safety profile and efficacy in eradicating CMV from patients, demonstrating its role as a standard treatment option .
Comparative Efficacy
| Study | Population | Treatment Comparison | Outcome |
|---|---|---|---|
| VICTOR Trial | Solid organ transplant recipients | Oral this compound vs. IV ganciclovir | Noninferior efficacy; similar success rates |
| Pediatric Pharmacokinetics | Children with CMV infection | This compound dosing adjustments | Effective treatment with adjusted dosing |
| Therapeutic Drug Monitoring Case | Lung transplant recipient | This compound prophylaxis | Successful viral suppression under monitoring |
Mechanism of Action
Valganciclovir is a prodrug that is rapidly converted to ganciclovir by hepatic and intestinal esterases . Ganciclovir is an acyclic analog of the nucleoside guanosine. Once inside the cell, ganciclovir is phosphorylated by viral kinases to form ganciclovir triphosphate . This triphosphate form inhibits viral DNA polymerase by competing with deoxyguanosine triphosphate, leading to the termination of viral DNA elongation . This mechanism effectively prevents the replication of cytomegalovirus and other herpesviruses .
Comparison with Similar Compounds
Ganciclovir: The active form of valganciclovir, used to treat cytomegalovirus infections.
Valacyclovir: An antiviral prodrug that is converted to acyclovir, used to treat herpes simplex virus infections.
Acyclovir: A nucleoside analog used to treat herpes simplex virus and varicella-zoster virus infections.
This compound stands out due to its effectiveness in treating cytomegalovirus infections and its ability to be administered orally, providing a convenient option for long-term treatment .
Biological Activity
Valganciclovir (VGC) is an antiviral medication primarily used for the prevention and treatment of cytomegalovirus (CMV) infections, particularly in immunocompromised patients such as those undergoing organ transplantation. This article delves into the biological activity of this compound, focusing on its pharmacological properties, clinical efficacy, and emerging research findings.
Pharmacological Properties
This compound is a prodrug of ganciclovir, which means it is converted into its active form after administration. The addition of a valine ester significantly enhances its oral bioavailability, making it a preferred option for outpatient therapy. The pharmacokinetics of VGC demonstrate that it achieves plasma concentrations comparable to those obtained with intravenous ganciclovir when administered at appropriate doses.
Key Pharmacokinetic Data
| Parameter | This compound (900 mg) | Ganciclovir (5 mg/kg IV) |
|---|---|---|
| Bioavailability | ~60% | ~6% |
| Peak Plasma Concentration | Achieved within 1-2 hours | Achieved within 1 hour |
| Half-life | 3-4 hours | 2-4 hours |
This enhanced bioavailability allows for effective outpatient management of CMV infections, reducing the need for intravenous therapy, which is often associated with higher costs and complications.
Clinical Efficacy
This compound has been shown to be effective in preventing and treating CMV disease in various patient populations, including organ transplant recipients and those with HIV. The VICTOR study established that oral this compound is noninferior to intravenous ganciclovir for treating CMV disease in solid organ transplant recipients, leading to its recommendation in clinical guidelines .
Case Studies and Clinical Trials
- VICTOR Study : This randomized trial compared oral this compound to intravenous ganciclovir in solid organ transplant recipients. It demonstrated comparable efficacy in eradicating CMV after 21 days of treatment and highlighted the importance of tailoring therapy based on initial viral load and immunosuppression levels .
- Phase I Trial in Idiopathic Pulmonary Fibrosis (IPF) : A recent study investigated this compound as an adjunct therapy in patients with IPF. Results indicated that while the primary endpoint was not met, there was a trend towards improved lung function metrics among those treated with this compound compared to placebo .
- Glioblastoma Treatment : Emerging research suggests that this compound may have potential applications beyond antiviral therapy. A study indicated that it improved survival rates in patients with newly diagnosed glioblastoma, particularly in those with unmethylated MGMT promoter genes. This finding opens avenues for further exploration into the drug's effects on tumor biology .
Safety Profile
The safety profile of this compound is critical, especially given its use in vulnerable populations. Common adverse effects include hematological toxicities such as leukopenia and neutropenia. A comparative analysis revealed that lower doses (450 mg) may be equally effective with a reduced risk of adverse events compared to higher doses (900 mg) .
Adverse Events Data
| Adverse Event | 450 mg VGC | 900 mg VGC |
|---|---|---|
| Leucopenia | 10% | 40% |
| Neutropenia | 5% | 20% |
| Discontinuation due to AE | 5% | 15% |
Q & A
Basic Research Questions
Q. What experimental models are most suitable for studying Valganciclovir’s antiviral efficacy against cytomegalovirus (CMV)?
- Methodological Answer : Use in vitro models such as human foreskin fibroblasts infected with CMV strains (e.g., AD169 or Towne) to quantify viral replication inhibition via plaque reduction assays. For in vivo studies, immunocompromised rodent models (e.g., murine CMV-infected mice) are preferred to simulate post-transplant scenarios. Dose-response relationships should be validated using pharmacokinetic (PK) parameters like AUC/MIC ratios .
Q. How can researchers address discrepancies in this compound’s bioavailability across patient subgroups?
- Methodological Answer : Conduct population pharmacokinetic (PopPK) analyses using nonlinear mixed-effects modeling (NONMEM) to identify covariates (e.g., renal function, weight) affecting bioavailability. Stratify data by subpopulations (e.g., pediatric vs. adult transplant recipients) and compare with historical ganciclovir data to adjust dosing algorithms .
Q. What are the validated analytical methods for quantifying this compound and its metabolites in biological samples?
- Methodological Answer : Reverse-phase high-performance liquid chromatography (RP-HPLC) with UV detection at 254 nm is widely used. Validate methods per ICH guidelines for linearity (5–50 µg/mL), precision (%RSD <2.0), and robustness (e.g., mobile phase ratios of 0.05% OPA:acetonitrile 20:80) . For low-concentration samples, LC-MS/MS offers higher sensitivity (LOQ: 0.1 ng/mL) .
Advanced Research Questions
Q. How should researchers design non-inferiority trials comparing this compound to alternative CMV prophylaxis regimens?
- Methodological Answer : Define a non-inferiority margin (Δ) based on historical data (e.g., Δ=10–15% for CMV disease incidence). Use intent-to-treat (ITT) and per-protocol (PP) analyses to mitigate bias, as ITT may dilute treatment effects. For example, Paya et al. (2004) used Δ=25% but faced criticism for potential type I errors; instead, employ adaptive trial designs with interim futility analyses .
Q. What strategies resolve contradictions in this compound’s cost-effectiveness across healthcare systems?
- Methodological Answer : Perform scenario analyses using Markov models to compare cost per QALY (quality-adjusted life year) in different settings. For instance, combining this compound with PET (preemptive therapy) reduced costs by $8,333/patient in the first year post-transplant. Sensitivity analyses should vary parameters like CMV serostatus prevalence and drug acquisition costs .
Q. How can genomic sequencing elucidate mechanisms of this compound resistance in CMV?
- Methodological Answer : Amplify UL97 (kinase) and UL54 (polymerase) gene regions from clinical isolates using PCR, followed by Sanger sequencing. Identify mutations (e.g., UL97 L595S) associated with reduced susceptibility. Correlate findings with phenotypic resistance via plaque reduction assays (IC₅₀ shifts ≥3-fold) .
Q. What ethical considerations arise when studying this compound’s teratogenic potential in preclinical models?
- Methodological Answer : Follow OECD 414 guidelines for embryo-fetal development studies in pregnant rats/rabbits. Dose ranges should mirror human exposures (AUC-based). For human data, use registries like the Antiretroviral Pregnancy Registry to track outcomes in HIV-positive pregnant women, acknowledging limitations in sample size and confounding factors .
Q. Data Synthesis and Validation
Q. How to systematically evaluate this compound’s long-term safety profile from heterogeneous clinical datasets?
- Methodological Answer : Conduct a meta-analysis using PRISMA guidelines. Pool adverse event data (e.g., neutropenia incidence) from RCTs and observational studies. Apply random-effects models to account for heterogeneity, and assess publication bias via funnel plots .
Q. What statistical approaches reconcile conflicting efficacy outcomes in this compound trials?
- Methodological Answer : Use Bayesian network meta-analysis to compare this compound against multiple comparators (e.g., ganciclovir, letermovir). Rank treatments via surface under the cumulative ranking curve (SUCRA), adjusting for trial quality (Jadad score) and patient characteristics .
Q. Tables for Key Findings
Properties
Key on ui mechanism of action |
Valganciclovir is an L-valyl ester (prodrug) of ganciclovir that exists as a mixture of two diastereomers. After oral administration, both diastereomers are rapidly converted to ganciclovir by intestinal and hepatic esterases. Ganciclovir is a synthetic analogue of 2'-deoxyguanosine, which inhibits replication of human CMV in cell culture and in vivo. In CMV-infected cells ganciclovir is initially phosphorylated to ganciclovir monophosphate by the viral protein kinase, pUL97. Further phosphorylation occurs by cellular kinases to produce ganciclovir triphosphate, which is then slowly metabolized intracellularly (half-life 18 hours). As the phosphorylation is largely dependent on the viral kinase, phosphorylation of ganciclovir occurs preferentially in virus-infected cells. The virustatic activity of ganciclovir is due to inhibition of the viral DNA polymerase, pUL54, synthesis by ganciclovir triphosphate. |
|---|---|
CAS No. |
175865-60-8 |
Molecular Formula |
C14H22N6O5 |
Molecular Weight |
354.36 g/mol |
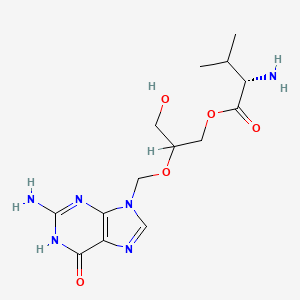
IUPAC Name |
[2-[(2-amino-6-oxo-1H-purin-9-yl)methoxy]-3-hydroxypropyl] 2-amino-3-methylbutanoate |
InChI |
InChI=1S/C14H22N6O5/c1-7(2)9(15)13(23)24-4-8(3-21)25-6-20-5-17-10-11(20)18-14(16)19-12(10)22/h5,7-9,21H,3-4,6,15H2,1-2H3,(H3,16,18,19,22) |
InChI Key |
WPVFJKSGQUFQAP-UHFFFAOYSA-N |
SMILES |
CC(C)C(C(=O)OCC(CO)OCN1C=NC2=C1N=C(NC2=O)N)N |
Isomeric SMILES |
CC(C)C(C(=O)OC[C@H](CO)OCN1C=NC2=C1N=C(NC2=O)N)N |
Canonical SMILES |
CC(C)C(C(=O)OCC(CO)OCN1C=NC2=C1N=C(NC2=O)N)N |
physical_description |
Solid |
Purity |
> 95% |
quantity |
Milligrams-Grams |
solubility |
4.79e+00 g/L |
Synonyms |
5-Amino-3-[1-(hydroxymethyl)-2-(L-valyloxy)ethoxymethyl]-6,7- dihydro-3H-imidazo[4,5-d]pyrimidin-7-one; (2S)-2-((2-AMino-6-oxo-1H-purin-9(6H)-yl)Methoxy)-3-hydroxypropyl 2-aMino-3-Methylbutanoate |
Origin of Product |
United States |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















