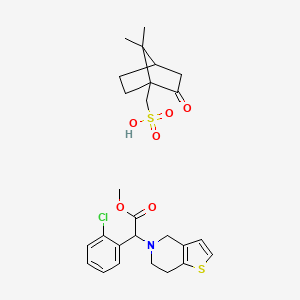
Clopidogrel Camphorsulfonate
- Klicken Sie auf QUICK INQUIRY, um ein Angebot von unserem Expertenteam zu erhalten.
- Mit qualitativ hochwertigen Produkten zu einem WETTBEWERBSFÄHIGEN Preis können Sie sich mehr auf Ihre Forschung konzentrieren.
Übersicht
Beschreibung
Clopidogrel Camphorsulfonate is a useful research compound. Its molecular formula is C26H32ClNO6S2 and its molecular weight is 554.1 g/mol. The purity is usually 95%.
BenchChem offers high-quality this compound suitable for many research applications. Different packaging options are available to accommodate customers' requirements. Please inquire for more information about this compound including the price, delivery time, and more detailed information at [email protected].
Wissenschaftliche Forschungsanwendungen
Pharmacological Properties
Clopidogrel is an inhibitor of platelet aggregation, primarily acting by blocking the binding of adenosine diphosphate (ADP) to its receptor on platelets. This mechanism reduces the likelihood of thrombus formation, which is crucial in preventing cardiovascular events such as myocardial infarction and stroke . The camphorsulfonate salt form enhances the stability and solubility of clopidogrel, making it suitable for pharmaceutical formulations .
Key Characteristics
- Stability : Clopidogrel camphorsulfonate exhibits improved stability against moisture and heat compared to other salt forms .
- Solubility : It has satisfactory water solubility, approximately 4.0 mg/ml, which facilitates its dissolution in pharmaceutical compositions .
- Optical Purity : The salt can achieve high optical purity (≥98.5% enantiomeric excess), crucial for ensuring therapeutic efficacy .
Clinical Applications
This compound is primarily indicated for the prevention of atherothrombotic events in various clinical scenarios:
- Acute Coronary Syndrome : It is used in combination with acetylsalicylic acid (ASA) for patients experiencing unstable angina or non-ST-segment elevation myocardial infarction .
- Post-Myocardial Infarction : Administered to prevent further thrombotic events within 35 days following an acute myocardial infarction .
- Ischemic Stroke : Effective in preventing recurrent strokes within six months after an initial event .
- Peripheral Arterial Disease : Used for patients with established peripheral arterial disease to reduce cardiovascular complications .
Efficacy in Post-PCI Patients
A study highlighted that patients undergoing percutaneous coronary intervention (PCI) benefit significantly from clopidogrel therapy, showing a relative risk reduction of 30-85% in major adverse cardiovascular events compared to placebo . This underscores the importance of this compound in high-risk populations.
Pharmacogenetic Considerations
Research indicates that genetic variations affecting the CYP2C19 enzyme can influence the efficacy of clopidogrel. Patients with loss-of-function alleles may experience reduced therapeutic benefits, emphasizing the need for genotype-guided therapy in specific populations .
Comparison with Other Antiplatelet Agents
Clopidogrel has been compared with other antiplatelet medications like ticagrelor and aspirin. Evidence suggests that clopidogrel remains a critical option for patients who may not tolerate newer agents due to side effects or contraindications .
Eigenschaften
Molekularformel |
C26H32ClNO6S2 |
|---|---|
Molekulargewicht |
554.1 g/mol |
IUPAC-Name |
(7,7-dimethyl-2-oxo-1-bicyclo[2.2.1]heptanyl)methanesulfonic acid;methyl 2-(2-chlorophenyl)-2-(6,7-dihydro-4H-thieno[3,2-c]pyridin-5-yl)acetate |
InChI |
InChI=1S/C16H16ClNO2S.C10H16O4S/c1-20-16(19)15(12-4-2-3-5-13(12)17)18-8-6-14-11(10-18)7-9-21-14;1-9(2)7-3-4-10(9,8(11)5-7)6-15(12,13)14/h2-5,7,9,15H,6,8,10H2,1H3;7H,3-6H2,1-2H3,(H,12,13,14) |
InChI-Schlüssel |
XEENARPWPCQXST-UHFFFAOYSA-N |
Kanonische SMILES |
CC1(C2CCC1(C(=O)C2)CS(=O)(=O)O)C.COC(=O)C(C1=CC=CC=C1Cl)N2CCC3=C(C2)C=CS3 |
Herkunft des Produkts |
United States |
Synthesis routes and methods
Procedure details





Haftungsausschluss und Informationen zu In-Vitro-Forschungsprodukten
Bitte beachten Sie, dass alle Artikel und Produktinformationen, die auf BenchChem präsentiert werden, ausschließlich zu Informationszwecken bestimmt sind. Die auf BenchChem zum Kauf angebotenen Produkte sind speziell für In-vitro-Studien konzipiert, die außerhalb lebender Organismen durchgeführt werden. In-vitro-Studien, abgeleitet von dem lateinischen Begriff "in Glas", beinhalten Experimente, die in kontrollierten Laborumgebungen unter Verwendung von Zellen oder Geweben durchgeführt werden. Es ist wichtig zu beachten, dass diese Produkte nicht als Arzneimittel oder Medikamente eingestuft sind und keine Zulassung der FDA für die Vorbeugung, Behandlung oder Heilung von medizinischen Zuständen, Beschwerden oder Krankheiten erhalten haben. Wir müssen betonen, dass jede Form der körperlichen Einführung dieser Produkte in Menschen oder Tiere gesetzlich strikt untersagt ist. Es ist unerlässlich, sich an diese Richtlinien zu halten, um die Einhaltung rechtlicher und ethischer Standards in Forschung und Experiment zu gewährleisten.















