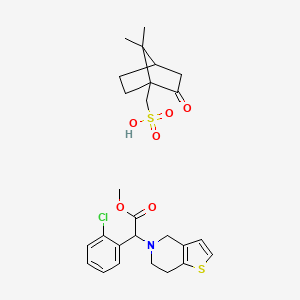
Clopidogrel Camphorsulfonate
- 専門家チームからの見積もりを受け取るには、QUICK INQUIRYをクリックしてください。
- 品質商品を競争力のある価格で提供し、研究に集中できます。
説明
Clopidogrel Camphorsulfonate is a useful research compound. Its molecular formula is C26H32ClNO6S2 and its molecular weight is 554.1 g/mol. The purity is usually 95%.
BenchChem offers high-quality this compound suitable for many research applications. Different packaging options are available to accommodate customers' requirements. Please inquire for more information about this compound including the price, delivery time, and more detailed information at [email protected].
科学的研究の応用
Pharmacological Properties
Clopidogrel is an inhibitor of platelet aggregation, primarily acting by blocking the binding of adenosine diphosphate (ADP) to its receptor on platelets. This mechanism reduces the likelihood of thrombus formation, which is crucial in preventing cardiovascular events such as myocardial infarction and stroke . The camphorsulfonate salt form enhances the stability and solubility of clopidogrel, making it suitable for pharmaceutical formulations .
Key Characteristics
- Stability : Clopidogrel camphorsulfonate exhibits improved stability against moisture and heat compared to other salt forms .
- Solubility : It has satisfactory water solubility, approximately 4.0 mg/ml, which facilitates its dissolution in pharmaceutical compositions .
- Optical Purity : The salt can achieve high optical purity (≥98.5% enantiomeric excess), crucial for ensuring therapeutic efficacy .
Clinical Applications
This compound is primarily indicated for the prevention of atherothrombotic events in various clinical scenarios:
- Acute Coronary Syndrome : It is used in combination with acetylsalicylic acid (ASA) for patients experiencing unstable angina or non-ST-segment elevation myocardial infarction .
- Post-Myocardial Infarction : Administered to prevent further thrombotic events within 35 days following an acute myocardial infarction .
- Ischemic Stroke : Effective in preventing recurrent strokes within six months after an initial event .
- Peripheral Arterial Disease : Used for patients with established peripheral arterial disease to reduce cardiovascular complications .
Efficacy in Post-PCI Patients
A study highlighted that patients undergoing percutaneous coronary intervention (PCI) benefit significantly from clopidogrel therapy, showing a relative risk reduction of 30-85% in major adverse cardiovascular events compared to placebo . This underscores the importance of this compound in high-risk populations.
Pharmacogenetic Considerations
Research indicates that genetic variations affecting the CYP2C19 enzyme can influence the efficacy of clopidogrel. Patients with loss-of-function alleles may experience reduced therapeutic benefits, emphasizing the need for genotype-guided therapy in specific populations .
Comparison with Other Antiplatelet Agents
Clopidogrel has been compared with other antiplatelet medications like ticagrelor and aspirin. Evidence suggests that clopidogrel remains a critical option for patients who may not tolerate newer agents due to side effects or contraindications .
特性
分子式 |
C26H32ClNO6S2 |
|---|---|
分子量 |
554.1 g/mol |
IUPAC名 |
(7,7-dimethyl-2-oxo-1-bicyclo[2.2.1]heptanyl)methanesulfonic acid;methyl 2-(2-chlorophenyl)-2-(6,7-dihydro-4H-thieno[3,2-c]pyridin-5-yl)acetate |
InChI |
InChI=1S/C16H16ClNO2S.C10H16O4S/c1-20-16(19)15(12-4-2-3-5-13(12)17)18-8-6-14-11(10-18)7-9-21-14;1-9(2)7-3-4-10(9,8(11)5-7)6-15(12,13)14/h2-5,7,9,15H,6,8,10H2,1H3;7H,3-6H2,1-2H3,(H,12,13,14) |
InChIキー |
XEENARPWPCQXST-UHFFFAOYSA-N |
正規SMILES |
CC1(C2CCC1(C(=O)C2)CS(=O)(=O)O)C.COC(=O)C(C1=CC=CC=C1Cl)N2CCC3=C(C2)C=CS3 |
製品の起源 |
United States |
Synthesis routes and methods
Procedure details





試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。














