Etoposide
Übersicht
Beschreibung
Etoposide ist ein Chemotherapeutikum, das hauptsächlich zur Behandlung verschiedener Krebsarten eingesetzt wird, darunter Hodenkrebs, Lungenkrebs, Lymphom, Leukämie, Neuroblastom und Eierstockkrebs . Es ist ein semisynthetisches Derivat von Podophyllotoxin, einem Naturstoff, der aus den Wurzeln und Rhizomen von Pflanzen der Gattung Podophyllum gewonnen wird . This compound wirkt, indem es das Enzym Topoisomerase II hemmt, das für die DNA-Replikation und Zellteilung unerlässlich ist .
Herstellungsmethoden
This compound wird durch einen semisynthetischen Prozess ausgehend von Podophyllotoxin synthetisiert. Eine gängige Methode beinhaltet die direkte Kondensation von 4’-Demethylepipodophyllotoxin mit 2,3-Di-O-Dichloracetyl-(4,6-O-Ethyliden)-β-D-Glucopyranose in Gegenwart von Trimethylsilyltrifluormethansulfonat (TMSOTf) zu 4’-Demethylepipodophyllotoxin-4-(2,3-Di-O-Dichloracetyl-4,6-0-Ethyliden)-β-D-Glucopyranosid, das dann in this compound umgewandelt wird . Diese Methode bietet im Vergleich zu bestehenden Techniken höhere Ausbeuten, verkürzte Reaktionszeiten und günstigere Isolierverfahren .
Vorbereitungsmethoden
Etoposide is synthesized through a semisynthetic process starting from podophyllotoxin. One common method involves the direct condensation of 4’-demethyl epipodophyllotoxin with 2,3-di-O-dichloroacetyl-(4,6-O-ethylidene)-β-D-glucopyranose in the presence of trimethylsilyl trifluoromethane sulfonate (TMSOTf) to yield 4’-demethylepipodophyllotoxin-4-(2,3-di-O-dichloroacetyl-4,6-0-ethylidene)-β-D-glucopyranoside, which is then converted to this compound . This method provides enhanced yields, reduced reaction times, and more favorable isolation procedures compared to existing techniques .
Analyse Chemischer Reaktionen
Etoposide unterliegt verschiedenen Arten von chemischen Reaktionen, darunter:
Reduktion: Reduktionsreaktionen können this compound in seine Hydrochinonform umwandeln.
Substitution: this compound kann Substitutionsreaktionen eingehen, insbesondere an der Glucopyranosid-Einheit.
Häufige Reagenzien und Bedingungen, die in diesen Reaktionen verwendet werden, umfassen Oxidationsmittel wie Wasserstoffperoxid und Reduktionsmittel wie Natriumborhydrid. Die Hauptprodukte, die aus diesen Reaktionen gebildet werden, umfassen die O-Chinon- und Hydrochinon-Derivate von this compound .
Wissenschaftliche Forschungsanwendungen
Etoposide is a semi-synthetic derivative of podophyllotoxin that inhibits topoisomerase-II, leading to DNA strand breaks and apoptosis . Since its FDA approval in 1983, this compound has been used to treat solid and hematologic tumors, including lung cancer, germ cell tumors, and lymphoma .
This compound in Other Cancer Treatments
This compound, often in combination with other chemotherapeutic agents, is used to treat various cancers .
- Non-Hodgkin's Lymphoma this compound, combined with lomustine, methotrexate, and prednisone, is a first-line therapy for non-Hodgkin's lymphoma without major cardiotoxicity .
- Hodgkin's Disease this compound is a first-line chemotherapeutic agent combined with vincristine, chloramphenicol, and prednisolone, with a 77% response rate .
- Brain Tumors this compound has been observed to work against brain tumors in combination therapy with cyclophosphamide, cisplatin, vincristine, and carboplatin .
- Breast Cancer this compound has been explored in the management of metastatic breast cancer . A single-agent trial in untreated patients showed a response rate of approximately 15% .
- Germ Cell Tumors Adjuvant this compound plus cisplatin for 2 cycles has demonstrated prolonged disease-specific and relapse-free survival with acceptable toxicity and lower drug costs in patients with nonseminomatous germ cell tumors .
- Choriocarcinoma Low-dose this compound and cisplatin (EP) can be effective in treating elderly patients with choriocarcinoma .
Adverse Events and Toxicity
Wirkmechanismus
Etoposide exerts its effects by inhibiting the enzyme topoisomerase II, which is responsible for regulating DNA topology through a double-strand passage mechanism . By forming a complex with topoisomerase II and DNA, this compound induces breaks in double-stranded DNA and prevents repair by topoisomerase II binding . Accumulated breaks in DNA prevent entry into the mitotic phase of cell division, leading to cell death . This compound acts primarily in the G2 and S phases of the cell cycle .
Vergleich Mit ähnlichen Verbindungen
Etoposide ähnelt anderen Podophyllotoxin-Derivaten, wie z. B. Teniposid und Podophyllotoxin selbst. This compound ist einzigartig in seiner spezifischen Hemmung der Topoisomerase II und seiner Verwendung in einer breiten Palette von Krebsbehandlungen .
Podophyllotoxin: Der Naturstoff, von dem this compound abgeleitet ist, wird hauptsächlich wegen seiner antiviralen und antimitotischen Eigenschaften eingesetzt.
Die Einzigartigkeit von this compound liegt in seiner semisynthetischen Natur, seiner verbesserten Stabilität und seinem breiten Spektrum an Antikrebsaktivität .
Biologische Aktivität
Etoposide is a semisynthetic derivative of podophyllotoxin, primarily utilized as an antineoplastic agent in cancer therapy. Its biological activity is predominantly characterized by its ability to inhibit DNA topoisomerase II, leading to significant effects on DNA synthesis and cell cycle progression. This article delves into the mechanisms of action, clinical applications, and recent research findings regarding this compound's biological activity.
This compound exerts its effects primarily through the following mechanisms:
- Inhibition of DNA Topoisomerase II : this compound forms a complex with topoisomerase II, preventing the re-ligation of DNA strands after they have been cleaved. This results in the accumulation of DNA double-strand breaks (DSBs), which can trigger apoptosis in cancer cells .
- Cell Cycle Specificity : The drug is cell cycle-dependent, affecting mainly the S and G2 phases. At high concentrations, it can induce cell lysis during mitosis, while lower concentrations inhibit cells from entering prophase .
- Activation of DNA Damage Response : this compound activates key proteins involved in the DNA damage response, such as ATM (Ataxia Telangiectasia Mutated) kinase, which leads to the formation of repair foci and chromosomal abnormalities if DSBs are not repaired .
Clinical Applications
This compound is widely used in various chemotherapy regimens for different types of cancers, particularly small-cell lung cancer (SCLC) and testicular cancer. Its effectiveness can be influenced by the scheduling of administration:
- Small-Cell Lung Cancer : In a randomized trial comparing two administration schedules, the five-day infusion regimen demonstrated an overall response rate of 89%, significantly higher than the 10% observed with a 24-hour continuous infusion schedule .
- Combination Therapies : this compound is often combined with other agents. For instance, in treating extensive SCLC, combinations with cisplatin have shown objective response rates ranging from 44% to 78% .
Research Findings and Case Studies
Recent studies have expanded our understanding of this compound's biological activity beyond its traditional uses:
- Antibacterial Activity : A study explored this compound's antibacterial properties when combined with hydroxyapatite, suggesting potential repurposing for treating infections .
- Pediatric Oncology : The OLIE trial evaluated the combination of lenvatinib, ifosfamide, and this compound in children with high-grade osteosarcoma, showing promising outcomes .
- Toxicity Management : Research has indicated that this compound can cause myelosuppressive toxicity; however, recent trials have focused on optimizing dosing regimens to mitigate these effects while maintaining efficacy .
Data Summary
The following table summarizes key findings from various studies on this compound:
Q & A
Basic Research Questions
Q. What is the primary mechanism by which etoposide exerts its anticancer effects?
this compound targets DNA topoisomerase II (Topo II), stabilizing the enzyme-DNA cleavage complex and preventing religation of DNA strands. This results in double-strand breaks (DSBs) and triggers apoptosis via activation of stress-associated signaling pathways (e.g., p53-mediated cell cycle arrest) . Notably, only 0.3% of this compound-induced strand breaks activate H2AX phosphorylation, suggesting most damage is non-toxic .
Q. How can researchers design experiments to assess this compound-induced cytotoxicity in vitro?
Standard protocols include measuring lactate dehydrogenase (LDH) release as a cell death marker, flow cytometry for cell cycle distribution (e.g., G2/M arrest), and apoptosis assays (Annexin V/PI staining). For example, SIN-1-treated astrocytes under glucose deprivation showed this compound's cytoprotective effect via LDH release quantification .
Q. What factors influence the variable oral bioavailability of this compound, and how can these be addressed preclinically?
this compound's low solubility (BCS Class IV) and rapid clearance (terminal half-life: 1.5 hours) limit bioavailability (24–74%). Researchers use nanosuspensions stabilized with surfactants (e.g., PLGA nanoparticles) and optimize formulations via Box–Behnken factorial designs to enhance dissolution and sustained release .
Advanced Research Questions
Q. How can experimental design mitigate biases in studies comparing this compound combination therapies (e.g., cisplatin + this compound)?
Rigorous protocols involve blinding, randomization, and standardized dosing. A meta-analysis of small-cell lung cancer trials highlighted the importance of controlling for confounders like patient stratification and toxicity metrics (e.g., myelosuppression rates) to validate efficacy claims .
Q. What molecular mechanisms underlie this compound resistance, and how can they be systematically investigated?
Resistance arises via reduced Topo II expression, overexpression of drug efflux pumps (ABCC1/ABCC6), or dysregulated signaling pathways (JAK-STAT, MAPK). Gene expression profiling (microarrays, RNA-seq) and functional assays (e.g., siRNA knockdown) in resistant cell lines (e.g., MCF7VP) reveal key pathways . Fluctuation analysis in MES-SA cells estimated mutation rates (2.9 × 10⁻⁶ to 1.7 × 10⁻⁷ per generation) for spontaneous resistance .
Q. How do contradictory findings on this compound's dual role in cytotoxicity and cytoprotection arise, and how can they be resolved?
this compound scavenges peroxynitrite (ONOO⁻) in glucose-deprived astrocytes, reducing oxidative stress independent of cell cycle inhibition . Contradictions may stem from model-specific variables (e.g., cell type, stress conditions). Researchers should validate mechanisms using multiple assays (e.g., glutathione depletion, superoxide anion measurement) .
Q. What advanced formulations improve this compound's pharmacokinetics, and how are they optimized?
Amorphous nanopowders (particle size: ~200 nm) using freeze-drying with cryoprotectants (mannitol) enhance oral absorption (Cmax: 2.21×; AUC: 2.13× vs. coarse powder). Optimization involves response surface methodology for ultrasonication time, phase ratios, and stabilizer concentration .
Q. Methodological Guidance
Q. How should researchers statistically analyze survival data from this compound-treated cohorts?
Use Kaplan-Meier survival curves with log-rank tests for significance. A meta-analysis of high-grade glioma trials reported median survival gains (SG: 7–23 months) and stratified results by treatment regimen (e.g., VP-16 vs. VM-26) .
Q. What in vitro models best recapitulate this compound's blood-brain barrier penetration for CNS cancer studies?
Primary rat astrocyte cultures under glucose deprivation and oxidative stress (SIN-1 exposure) mimic the blood-brain barrier's metabolic constraints. Measure NO release with Clark-type electrodes and superoxide with lucigenin chemiluminescence .
Q. How can researchers validate this compound's target engagement in DNA damage assays?
Comet assays quantify DSBs/SSBs, while γ-H2AX immunofluorescence marks repair foci. Calicheamicin-induced DSBs serve as positive controls to distinguish Topo II-dependent damage .
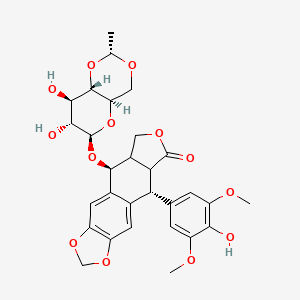
Eigenschaften
CAS-Nummer |
33419-42-0 |
|---|---|
Molekularformel |
C29H32O13 |
Molekulargewicht |
588.6 g/mol |
IUPAC-Name |
(5S,5aR,8aR,9R)-5-[[(2R,4aR,6R,7S,8R,8aS)-7,8-dihydroxy-2-methyl-4,4a,6,7,8,8a-hexahydropyrano[3,2-d][1,3]dioxin-6-yl]oxy]-9-(4-hydroxy-3,5-dimethoxyphenyl)-5a,6,8a,9-tetrahydro-5H-[2]benzofuro[6,5-f][1,3]benzodioxol-8-one |
InChI |
InChI=1S/C29H32O13/c1-11-36-9-20-27(40-11)24(31)25(32)29(41-20)42-26-14-7-17-16(38-10-39-17)6-13(14)21(22-15(26)8-37-28(22)33)12-4-18(34-2)23(30)19(5-12)35-3/h4-7,11,15,20-22,24-27,29-32H,8-10H2,1-3H3/t11-,15+,20-,21-,22+,24-,25+,26-,27-,29+/m1/s1 |
InChI-Schlüssel |
VJJPUSNTGOMMGY-QBUITQBFSA-N |
Verunreinigungen |
The following impurities are limited by the requirements of The British Pharmacopoeia: 4'-carbenzoxy ethylidene lignan P, picroethylidene lignan P, alpha-ethylidene lignan P, lignan P and 4'-demethylepipodophyllotoxin. |
SMILES |
CC1OCC2C(O1)C(C(C(O2)OC3C4COC(=O)C4C(C5=CC6=C(C=C35)OCO6)C7=CC(=C(C(=C7)OC)O)OC)O)O |
Isomerische SMILES |
C[C@@H]1OC[C@@H]2[C@@H](O1)[C@@H]([C@@H]([C@@H](O2)O[C@H]3[C@H]4COC(=O)[C@@H]4[C@@H](C5=CC6=C(C=C35)OCO6)C7=CC(=C(C(=C7)OC)O)OC)O)O |
Kanonische SMILES |
CC1OCC2C(O1)C(C(C(O2)OC3C4COC(=O)C4C(C5=CC6=C(C=C35)OCO6)C7=CC(=C(C(=C7)OC)O)OC)O)O |
Aussehen |
White to off-white solid powder |
Color/Form |
Crystals from methanol |
melting_point |
236-251 °C |
Key on ui other cas no. |
33419-42-0 |
Physikalische Beschreibung |
Solid |
Piktogramme |
Irritant; Health Hazard |
Reinheit |
>98% (or refer to the Certificate of Analysis) |
Haltbarkeit |
>2 years if stored properly |
Löslichkeit |
Very soluble in methanol, chloroform; slightly soluble in ethanol, sparingly soluble in water. Sol in alc: approx 0.76 mg/ml Water solubility: approx 0.08 mg/mL |
Lagerung |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
Synonyme |
alpha-D-Glucopyranosyl Isomer Etoposide Celltop Demethyl Epipodophyllotoxin Ethylidine Glucoside Eposide Eposin Eto GRY Eto-GRY Etomedac Etopos Etoposide Etoposide Pierre Fabre Etoposide Teva Etoposide, (5a alpha)-Isomer Etoposide, (5a alpha,9 alpha)-Isomer Etoposide, (5S)-Isomer Etoposide, alpha D Glucopyranosyl Isomer Etoposide, alpha-D-Glucopyranosyl Isomer Etoposido Ferrer Farma Exitop Lastet NSC 141540 NSC-141540 NSC141540 Onkoposid Riboposid Teva, Etoposide Toposar Vépéside Sandoz Vépéside-Sandoz Vepesid VP 16 VP 16 213 VP 16-213 VP 16213 VP-16 VP16 |
Dampfdruck |
5.4X10-23 mm Hg at 25 °C /Estimated/ |
Herkunft des Produkts |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Haftungsausschluss und Informationen zu In-Vitro-Forschungsprodukten
Bitte beachten Sie, dass alle Artikel und Produktinformationen, die auf BenchChem präsentiert werden, ausschließlich zu Informationszwecken bestimmt sind. Die auf BenchChem zum Kauf angebotenen Produkte sind speziell für In-vitro-Studien konzipiert, die außerhalb lebender Organismen durchgeführt werden. In-vitro-Studien, abgeleitet von dem lateinischen Begriff "in Glas", beinhalten Experimente, die in kontrollierten Laborumgebungen unter Verwendung von Zellen oder Geweben durchgeführt werden. Es ist wichtig zu beachten, dass diese Produkte nicht als Arzneimittel oder Medikamente eingestuft sind und keine Zulassung der FDA für die Vorbeugung, Behandlung oder Heilung von medizinischen Zuständen, Beschwerden oder Krankheiten erhalten haben. Wir müssen betonen, dass jede Form der körperlichen Einführung dieser Produkte in Menschen oder Tiere gesetzlich strikt untersagt ist. Es ist unerlässlich, sich an diese Richtlinien zu halten, um die Einhaltung rechtlicher und ethischer Standards in Forschung und Experiment zu gewährleisten.















