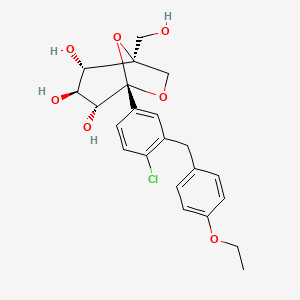
Ertugliflozin
描述
恩格列净是一种选择性钠-葡萄糖协同转运体2 (SGLT2) 抑制剂,主要用于治疗2型糖尿病。它通过阻断肾脏中葡萄糖的重吸收起作用,从而导致尿液中葡萄糖排泄增加。 这有助于降低 2 型糖尿病患者的血糖水平 .
准备方法
合成路线和反应条件: 恩格列净的合成涉及多个步骤,从用三苯甲基保护中间体化合物中伯醇部分开始,在吡啶存在下进行。 然后用对甲苯磺酸进行脱保护 . 该过程还涉及使用具有式 III、式 IV 和式 VII 的化合物,这些化合物用合适的保护基进行保护,以确保高纯度和高收率 .
工业生产方法: 恩格列净的工业生产遵循类似的合成路线,但规模更大。该过程涉及使用先进的技术和设备,以确保高收率和高纯度。 尽量减少使用吡啶等危险化学品,以确保安全并符合工业标准 .
化学反应分析
Degradation Reactions Under Stress Conditions
Ertugliflozin exhibits stability under thermal, photolytic, neutral, and alkaline hydrolysis conditions but degrades in acidic and oxidative environments. These reactions were studied as per ICH guidelines (Q1A–Q1E) to identify degradation products (DPs) and their mechanisms .
Key Degradation Pathways:
-
Acid Hydrolysis (1N HCl at 60°C for 48 h) :
- Forms four degradation products (DP-1 to DP-4) via:
- Ritter reaction (DP-1: acetonitrile reacts with protonated formaldehyde).
- Benzyl ether cleavage (DP-2: acid-mediated elimination of the ethoxybenzyl group).
- Esterification (DP-3: reaction with acetic acid from sugar decomposition).
- Chlorination (DP-4: SN2 displacement of hydroxymethyl group by chloride) .
- Forms four degradation products (DP-1 to DP-4) via:
- Oxidative Hydrolysis (30% H₂O₂ at RT for 48 h) :
Structural Characterization of Degradation Products
All five degradation products were characterized using UHPLC-MS , HRMS , NMR (1D/2D) , and IR spectroscopy . Key data include:
| Product | Molecular Formula | m/z (Observed) | Key Structural Features |
|---|---|---|---|
| DP-1 | C₂₅H₃₀ClNO₈ | 508.1724 | Benzylacetamide derivative |
| DP-2 | C₂₁H₁₉ClO₄ | 371.1037 | De-ethoxybenzyl analog |
| DP-5 | C₂₀H₁₇ClO₆ | 413.1139 | Phenolic oxidative product |
Metabolic Reactions
This compound is primarily metabolized via glucuronidation (86%) and minor oxidative pathways (12%) :
- UGT1A9/UGT2B7-mediated glucuronidation : Forms inactive metabolites M5a (2-O-β-glucuronide) and M5c (3-O-β-glucuronide).
- CYP3A4-mediated oxidation : Produces hydroxy (M1/M3) and desethyl (M2) metabolites .
Synthetic Reactions
The commercial synthesis of this compound involves:
- Cocrystallization : this compound is stabilized as a 1:1 cocrystal with L-pyroglutamic acid to enhance solubility and stability .
- Key intermediates : Protection/deprotection steps using formaldehyde and acetonitrile under acidic conditions .
Stability in Formulations
- pH-dependent solubility : Sparingly soluble in water (0.64–0.74 mg/mL at physiological pH) .
- Humidity-induced dissociation : The cocrystal partially converts to amorphous this compound under high humidity but remains bioequivalent .
Interaction with Analytical Reagents
This compound reacts with formaldehyde and dichloromethane during stress testing, leading to DP-1 and DP-4 formation .
科学研究应用
Clinical Efficacy in Type 2 Diabetes Management
Ertugliflozin has demonstrated significant reductions in HbA1c levels across various studies. In a pivotal phase 3 trial, participants showed a mean reduction in HbA1c of 0.6% to 0.9% compared to placebo after 52 weeks of treatment .
Key Clinical Trials
- VERTIS Trials : These trials evaluated the efficacy and safety of this compound in patients with T2DM and established cardiovascular disease. The results indicated that this compound was noninferior to placebo concerning major adverse cardiovascular events, but it did show trends toward reduced hospitalization for heart failure .
- Long-term Efficacy : A study assessing long-term outcomes reported sustained reductions in HbA1c over two years, with patients on higher doses (15 mg) showing more significant improvements compared to lower doses (5 mg) .
Cardiovascular Benefits
This compound has been associated with cardiovascular benefits beyond glycemic control. The VERTIS CV trial highlighted that this compound reduced the risk of hospitalization for heart failure by approximately 30% compared to placebo .
Cardiovascular Outcomes Summary
| Outcome Measure | This compound (5 mg) | This compound (15 mg) | Placebo |
|---|---|---|---|
| Major Adverse Cardiovascular Events | Noninferior | Noninferior | Reference |
| Hospitalization for Heart Failure | 2.5% | 2.8% | 3.6% |
| CV Death | 1.8% | 1.9% | 1.9% |
Renal Outcomes
Recent studies have also explored the renal protective effects of this compound. It has shown promise in improving renal outcomes in patients with chronic kidney disease (CKD) and T2DM, demonstrating a reduction in the progression of kidney disease markers .
Renal Outcomes Summary
| Renal Outcome Measure | This compound | Placebo |
|---|---|---|
| Doubling of Serum Creatinine | Reduced incidence | Higher incidence |
| Need for Dialysis/Transplantation | Lower rates | Higher rates |
Safety Profile
While this compound is generally well-tolerated, some adverse effects have been noted, including an increased risk of genital mycotic infections and urinary tract infections . However, these side effects are manageable and do not outweigh the benefits seen in glycemic control and cardiovascular health.
Combination Therapy
This compound can be used as monotherapy or in combination with other antidiabetic agents such as metformin or sitagliptin. Clinical trials have shown that such combinations can lead to enhanced glycemic control while minimizing the risk of hypoglycemia associated with insulin or sulfonylureas .
作用机制
恩格列净通过抑制肾脏中的钠-葡萄糖协同转运体 2 (SGLT2) 发挥作用。 这种抑制阻止了肾小球滤液中葡萄糖的重吸收,导致尿液中葡萄糖排泄增加,血葡萄糖水平降低 . 参与此过程的分子靶标包括位于肾脏近端小管中的 SGLT2 蛋白 .
相似化合物的比较
恩格列净是 SGLT2 抑制剂类药物的一部分,其中还包括卡格列净、达格列净和恩格列净。 与这些化合物相比,恩格列净在降低血糖水平方面显示出类似的疗效,但其安全性 and 副作用可能不同 . 例如,已发现恩格列净比其他一些 SGLT2 抑制剂导致尿路感染的风险更低 .
类似化合物列表:- 卡格列净
- 达格列净
- 恩格列净
- 贝格列净
生物活性
Ertugliflozin is a sodium-glucose cotransporter-2 (SGLT2) inhibitor that plays a significant role in the management of type 2 diabetes mellitus (T2DM). This compound enhances glycemic control by promoting urinary glucose excretion, thereby lowering blood glucose levels. This article delves into the biological activity of this compound, highlighting its pharmacokinetics, mechanisms of action, clinical efficacy, and safety profile based on diverse research findings.
This compound selectively inhibits SGLT2, a protein responsible for the reabsorption of glucose in the kidneys. By blocking this transporter, this compound increases urinary glucose excretion and decreases blood glucose levels. The selectivity of this compound for SGLT2 over SGLT1 is significant, with an IC50 value of 0.877 nM for SGLT2 compared to 1960 nM for SGLT1, indicating a greater than 2000-fold selectivity .
Pharmacokinetics
This compound exhibits favorable pharmacokinetic properties:
- Bioavailability : Approximately 100% when administered orally.
- Peak Concentration : Achieved within 1-2 hours post-dose.
- Half-life : Approximately 16.6 hours in T2DM patients, supporting once-daily dosing.
- Metabolism : Primarily metabolized by UGT1A9 and UGT2B7 through O-glucuronidation into inactive metabolites .
Clinical Efficacy
This compound has been evaluated in multiple clinical trials assessing its efficacy as monotherapy and in combination with other antidiabetic agents. Key findings include:
- VERTIS-CV Trial : Demonstrated a reduction in hospitalizations for heart failure among patients treated with this compound compared to placebo .
- Long-term Studies : A 104-week study showed significant reductions in HbA1c levels (up to -0.84% at week 104) and improvements in fasting plasma glucose (FPG) and body weight .
Table 1: Summary of Key Clinical Trials Involving this compound
| Trial Name | Population | Duration | Outcome Measures | Key Findings |
|---|---|---|---|---|
| VERTIS-CV | Patients with T2DM & CVD | 3.5 years | Hospitalizations for heart failure | Reduced hospitalizations vs placebo |
| Phase III Study | T2DM patients inadequately controlled on metformin | 104 weeks | HbA1c, FPG, body weight | HbA1c reduction up to -0.84% |
| Efficacy Study | Various T2DM populations | Varies | HbA1c, FPG | Significant reductions compared to placebo |
Safety Profile
While this compound is generally well-tolerated, it is associated with some adverse effects:
- Genital Mycotic Infections : Higher incidence reported among patients treated with this compound compared to placebo.
- Diabetic Ketoacidosis : Rare cases have been documented, particularly in patients with underlying conditions such as Latent Autoimmune Diabetes of Adulthood (LADA) .
- Bone Mineral Density : Minimal changes observed; however, some reductions were noted at the hip region after long-term use .
Case Studies
Several case studies have highlighted the real-world efficacy and safety of this compound:
- Case Study on Cardiovascular Outcomes : A patient with established cardiovascular disease showed improved cardiovascular outcomes after initiating treatment with this compound alongside standard diabetes management.
- Long-term Management Case : A patient inadequately controlled on metformin alone achieved target HbA1c levels after adding this compound to their regimen over a period of six months.
常见问题
Basic Research Questions
Q. What are the primary efficacy endpoints of ertugliflozin in phase III clinical trials, and how were they statistically validated?
this compound demonstrated clinically meaningful reductions in HbA1c, body weight, and systolic blood pressure across the VERTIS phase III trials (e.g., VERTIS CV, VERTIS SU, VERTIS MET). These outcomes were assessed using mixed-model repeated measures (MMRM) and last observation carried forward (LOCF) imputation for missing data. For example, in VERTIS SU, placebo-adjusted HbA1c reductions were −0.69% (5 mg) and −0.76% (15 mg) at 18 weeks, with p < 0.001 . Secondary endpoints like fasting plasma glucose and blood pressure changes were analyzed using logistic regression and log-rank tests for time-to-event outcomes .
Q. How was cardiovascular safety established for this compound in high-risk type 2 diabetes (T2DM) populations?
The VERTIS CV trial (N=8,246) demonstrated non-inferiority of this compound versus placebo for major adverse cardiovascular events (MACE; HR 0.97, 95.6% CI 0.85–1.11). The trial design included a double-blind, placebo-controlled protocol with pooled 5 mg/15 mg dosing and a pre-specified non-inferiority margin of 1.3. Secondary outcomes, such as cardiovascular death or heart failure hospitalization, showed a non-significant trend toward benefit (HR 0.88, 95.8% CI 0.75–1.03) .
Q. What methodologies are used to evaluate this compound’s renal effects in long-term studies?
Post hoc analyses of pooled data from VERTIS MET and VERTIS SU trials (N=1,237) assessed renal outcomes over 104 weeks. Changes in estimated glomerular filtration rate (eGFR) and urinary albumin-to-creatinine ratio (UACR) were analyzed using linear mixed-effects models. This compound showed a 19% relative risk reduction in composite renal endpoints (e.g., eGFR decline, ESKD) compared to non-ertugliflozin treatments, with adjustments for baseline covariates like UACR category .
Advanced Research Questions
Q. How do contradictory findings between cardiovascular and renal outcomes inform mechanistic hypotheses for this compound?
While this compound did not show superiority in MACE reduction, its renal benefits (e.g., 19% risk reduction in composite renal endpoints) suggest distinct pathways. Mediation analyses from empagliflozin trials propose mechanisms such as hematocrit elevation (indicating plasma volume reduction) and metabolic effects (e.g., uric acid reduction). These findings highlight the need for targeted studies on hemodynamic versus metabolic mediators in this compound’s organ protection .
Q. What statistical approaches address missing data in this compound trials, and how do they impact validity?
Trials like VERTIS SU used multiple imputation based on a constrained longitudinal data analysis (cLDA) model for missing HbA1c data at week 18. Sensitivity analyses comparing LOCF and MMRM methods confirmed robustness, with consistent HbA1c reductions (placebo-adjusted LS mean: −0.6% to −0.7%) across methodologies . However, residual bias from unmeasured confounders in imputation models remains a limitation .
Q. How do subgroup analyses by baseline HbA1c, age, or eGFR influence the interpretation of this compound’s efficacy?
In VERTIS CV sub-studies, this compound’s HbA1c reductions were consistent across subgroups (e.g., baseline HbA1c ≥8.5%: −0.8% vs. placebo). However, in patients with eGFR <60 mL/min/1.73 m², the glycemic effect attenuated, necessitating stratified dosing recommendations. These analyses used interaction tests with Bonferroni correction to control for Type I error .
Q. What evidence supports or refutes the association between this compound and amputations?
Pooled phase III data reported amputation rates of 2.0% (5 mg) and 2.1% (15 mg) versus 1.6% for placebo, with exposure-adjusted incidence rates of 5.0–6.8 vs. 4.3 per 1,000 patient-years. However, the VERTIS CV trial found no statistically significant increase (HR 1.14, 95% CI 0.75–1.74), suggesting confounding by baseline risk factors (e.g., peripheral artery disease) .
Q. Methodological Considerations
Q. How are composite renal endpoints defined and validated in this compound trials?
Renal endpoints in VERTIS trials included a clinically relevant hierarchy: ≥40% eGFR decline, end-stage kidney disease (ESKD), or renal death. These were adjudicated by blinded endpoint committees using standardized criteria. Sensitivity analyses excluding non-adjudicated events confirmed consistency in effect sizes .
Q. What are the limitations of indirect treatment comparisons (ITCs) for this compound versus other SGLT2 inhibitors?
ITCs in the 2018 PBAC report highlighted heterogeneity in trial designs (e.g., background therapies, follow-up duration) and inappropriate handling of multi-arm trials. For example, this compound showed non-inferiority to dapagliflozin in HbA1c reduction but had wider CIs due to fewer trials, limiting reliability .
Q. How do post hoc and prespecified analyses differ in interpreting this compound’s heart failure outcomes?
Prespecified analyses in VERTIS CV showed no significant reduction in heart failure hospitalization (HR 0.88, p=0.11), but sensitivity analyses using fixed-effect models suggested clinically meaningful benefits. This discrepancy underscores the importance of pre-registered statistical plans to avoid Type I error inflation .
属性
IUPAC Name |
(1S,2S,3S,4R,5S)-5-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H25ClO7/c1-2-28-16-6-3-13(4-7-16)9-14-10-15(5-8-17(14)23)22-20(27)18(25)19(26)21(11-24,30-22)12-29-22/h3-8,10,18-20,24-27H,2,9,11-12H2,1H3/t18-,19-,20+,21-,22-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
MCIACXAZCBVDEE-CUUWFGFTSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCOC1=CC=C(C=C1)CC2=C(C=CC(=C2)C34C(C(C(C(O3)(CO4)CO)O)O)O)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCOC1=CC=C(C=C1)CC2=C(C=CC(=C2)[C@@]34[C@@H]([C@H]([C@@H]([C@@](O3)(CO4)CO)O)O)O)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H25ClO7 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID40153120 | |
| Record name | PF-04971729 | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40153120 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
436.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Very slightly soluble | |
| Record name | Ertugliflozin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11827 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
As part of a normal process, the glucose from the blood is filtered for excretion and reabsorbed in the glomerulus so less than one percent of this glucose is excreted in the urine. The reabsorption is mediated by the sodium-dependent glucose cotransporter (SGLT), mainly the type 2 which is responsible for 90% of the reabsorbed glucose. Ertugliflozin is a small inhibitor of the SGLT2 and its activity increases glucose excretion, reducing hyperglycemia without the requirement of excessive insulin secretion. | |
| Record name | Ertugliflozin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11827 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1210344-57-2, 1431329-06-4, 1210344-83-4 | |
| Record name | Ertugliflozin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1210344-57-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Ertugliflozin [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1210344572 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Ertugliflozin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11827 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PF-04971729 | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40153120 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 1,6-Anhydro-1-C-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-5-C-(hydroxymethyl)-beta-L-idopyranose | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Ertugliflozin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Ertugliflozin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Ertugliflozin | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/6C282481IP | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。















